How to Read a Supplement COA (Certificate of Analysis) Without a Science Degree
Introduction
If you’ve been selling supplements for any length of time, you’ve probably received a COA from your supplier and done that thing where you nod like you understand it, then quietly googled “what does ND mean on a lab report” at 11 PM.
You’re not alone. Most sellers never actually read these documents — they just forward them to customers to look professional.
But here’s the problem: a COA is the only proof that what’s in the bottle matches what’s on the label. If you’re importing supplements and you can’t read the document your supplier hands you, you’re essentially flying blind.
This guide will teach you the five things on any COA that actually matter — no chemistry degree required.
What Is a COA and Why Should You Care?
A Certificate of Analysis (COA) is a lab report issued by a third-party testing facility that verifies the contents of a product batch. It’s not the same as a supplement facts label — it’s raw data.
What a COA tells you:
- What’s actually in the product (vs. what’s on the label)
- Whether it contains harmful contaminants
- That the potency matches what the manufacturer claims
- That the batch is safe to sell
Why most sellers ignore it:
It looks like a wall of numbers with abbreviations that feel like they were designed to confuse. Plus, your supplier probably told you it’s “all good” — so why dig in?
Because “all good” is not a lab result.
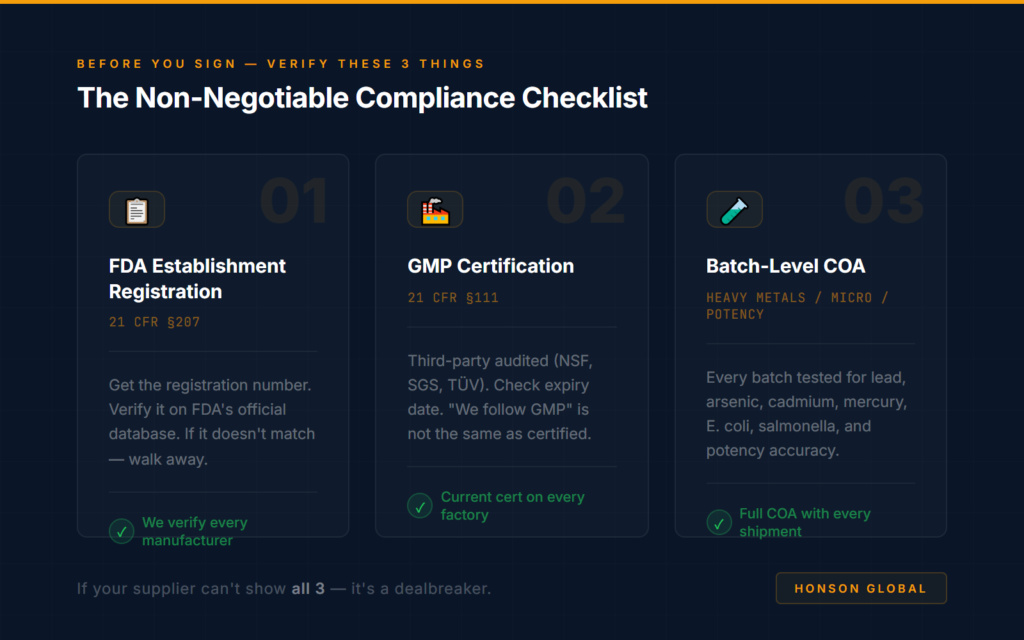
The Five Things That Actually Matter on Any COA
Here’s what to look for. Everything else on the document is context — these five sections are the actual decision points.
2.1 — Sample Information / Batch ID
Look for: Batch number, manufacturing date, sample date, testing date.
This is your audit trail. If a customer reports a problem, you need to trace back to the exact batch. A COA without a batch number is useless for recalls.
⚠️ Red flag: The batch number on your COA doesn’t match the batch on your product label.
2.2 — Potency / Assay Results
Look for: Active ingredient name, test method, result, unit.
Example: “Vitamin D3 — 5000 IU — HPLC — 4987 IU” means the lab tested for D3 using the HPLC method and found 4987 IU — within acceptable variance of 5000.
Acceptable variance: ±10% is standard. Some stricter labs use ±5%.
⚠️ Red flag: Potency is listed as “pass” with no actual number. Numbers matter — not labels.
2.3 — Contaminant Testing (Heavy Metals)
Look for: Lead, Arsenic, Cadmium, Mercury.
| Contaminant | FDA DRL (μg/day) |
|---|---|
| Lead | 2.5 |
| Arsenic (inorganic) | Not established (litigation risk) |
| Cadmium | 4.1 |
| Mercury | 2.0 |
“ND” means “Not Detected” — the lab tested and found nothing above the detection limit. This is good. ND is not the same as zero — it’s “the instrument can’t see it” — but it’s the best result you can get.
⚠️ Red flag: Heavy metals section is completely missing. Walk away.

2.4 — Microbiological Testing
Look for: E. coli, Salmonella, Total Plate Count, Yeast & Mold.
Contamination can happen at any point — sourcing, manufacturing, packaging, shipping. Micro testing confirms the product is shelf-stable and safe.
⚠️ Red flag: No micro testing section at all. Especially critical for probiotic products.
2.5 — The Testing Lab’s Accreditation
Look for: ISO 17025, lab name, lab accreditation number.
This is the most overlooked section. Any lab can run a test. Only accredited labs produce legally defensible results. ISO 17025 is the international standard for testing and calibration laboratories.
⚠️ Red flag: COA doesn’t list an accredited lab — the results are essentially worthless.
What a Real COA Looks Like — Side-by-Side
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Good COA includes:
- Full batch identification
- Accredited third-party lab name + ISO 17025 number
- Potency results with actual numerical values
- Heavy metals: all four metals tested with results
- Micro testing: all four categories
- Test method listed for each parameter (HPLC, ICP-MS, etc.)
- Date of testing and date of report
Bad COA includes:
- Supplier’s own logo (not a third-party lab)
- No lab name, no accreditation number
- “Pass” or “Complies” with no numbers
- Missing heavy metals section entirely
- Batch number doesn’t match product label
- Report date older than the batch manufacturing date
How to Request a COA From Your Supplier
Most legitimate manufacturers will have COAs ready for every batch. Here’s the right way to ask:
Hi [Supplier Name],
Before we place our order for [Product + Batch Size], we’d like to review the Certificate of Analysis for your most recent production run. Specifically, we need:
- Potency results (active ingredient assay)
- Heavy metals panel (lead, arsenic, cadmium, mercury)
- Microbiological testing
- Third-party lab name and ISO 17025 accreditation number
If the testing was done in-house rather than by an accredited third-party lab, please let us know — we may need to arrange independent testing before shipping.
Thanks,
[Your Name / Company Name]
Legitimate manufacturers will send this within 24-48 hours. If they push back, deflect, or send something that doesn’t include all five sections — that’s your answer.
The One Question That Reveals Everything
If you only remember one thing from this article, remember this question:
“Can I verify this COA independently — at the lab listed on the document?”
If your supplier hesitates, says “our lab doesn’t take outside inquiries,” or the lab name on the COA doesn’t appear in any public directory — you have your answer.
Final Thoughts
COAs are not complicated. The science behind them is complex, but the document itself has a clear job: tell you what’s in the product. Once you know what to look for, you can read any COA in under five minutes.
The sellers who get burned are the ones who never looked.
Need a supplier you can trust?
Honson Global sources every product from FDA-registered, GMP-certified manufacturers with full batch-level COA documentation — before your order ever ships.