The Supplement Compliance Mistake Most New Brands Are Making (And How to Fix It)
Intro
Last month, a founder reached out to us after losing his Amazon seller account.
His mistake? He spent $8,500 on 5,000 bottles of turmeric capsules — and his supplier never bothered with FDA facility registration or GMP certification. One customer complaint about “off-smell” capsules triggered an FDA inquiry, and within two weeks, his listing was suspended.
He’s not alone. We’ve seen this exact scenario play out at least 20 times in the past year alone.
The supplement market is booming — projected to hit $230 billion by 2027. But here’s the uncomfortable truth most “how to start a supplement brand” guides won’t tell you:
The #1 reason new supplement brands fail isn’t bad marketing or poor product quality. It’s compliance shortcuts taken before day one.
The Mistake: Trusting Your Supplier’s Word on Compliance
Here’s what happens to most first-time founders:
- You find a supplier on Alibaba or through a referral
- They say “yes, we’re FDA registered and GMP certified”
- You take their word for it and place your first order
- Everything seems fine — until it isn’t
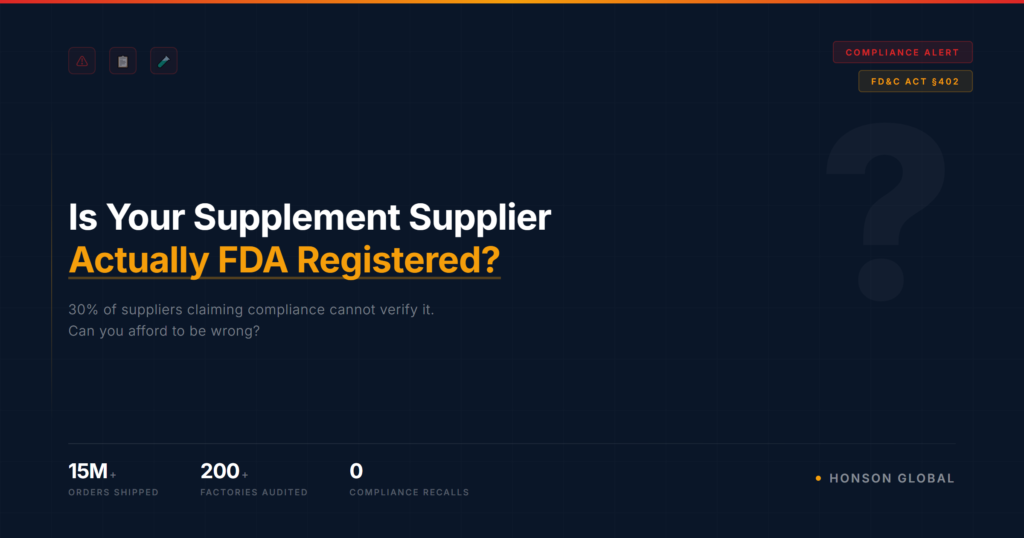
The hard truth: at least 30% of the suppliers claiming FDA registration on Alibaba cannot verify it on the official FDA database.
We know this because we’ve audited over 200 factories in Shenzhen, Guangzhou, and Yiwu over the past 20 years. Many of them have impressive websites and convincing sales teams. But when you ask for the actual FDA establishment registration number and cross-check it on FDA’s official database? Nothing.
No registration. No GMP audit records. No COA (Certificate of Analysis) for raw materials.
Your product is non-compliant the moment it enters US soil. You just don’t know it yet.
What This Actually Costs You
This isn’t a theoretical risk. Here’s what non-compliance looks like in real dollars:
Scenario
Real Cost
Amazon listing suspended
$5,000–$15,000 in lost revenue per week
FDA warning letter
$100,000+ (manufacturing + logistics + disposal)
Product recall
$100,000+ (manufacturing + logistics + disposal)
Customer injury lawsuit
$500,000–$5,000,000+
Brand reputation damage
Unquantifiable — often fatal for new brands

One compliance shortcut can wipe out your entire investment in a single afternoon.
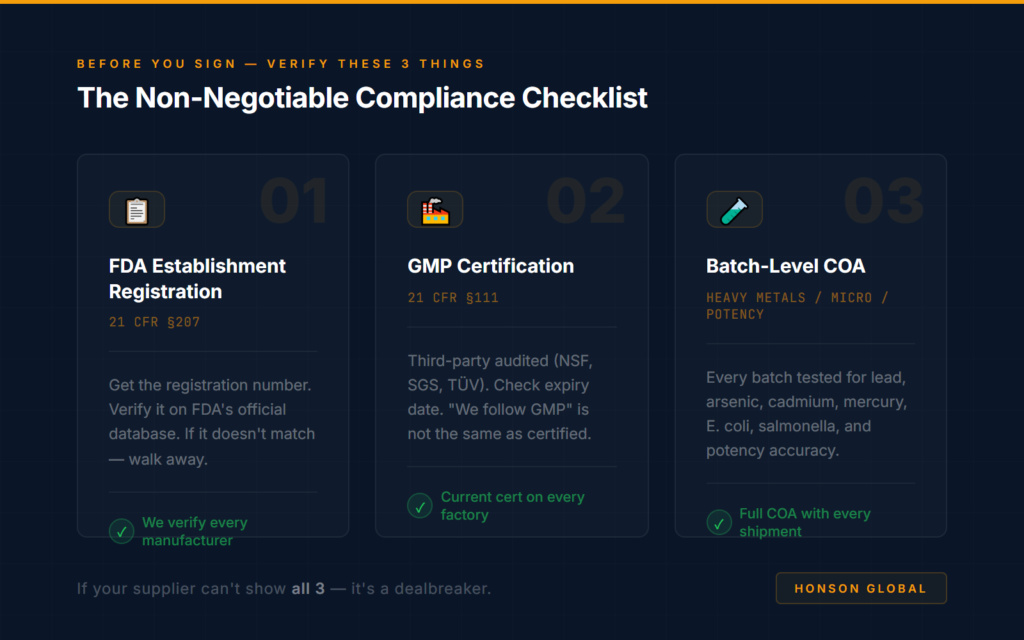
The Fix: 3 Things to Verify Before Signing With Any Supplier
Don’t just ask “are you compliant?” — ask for proof:
1. Regulatory Compliance is the Baseline
- Partner factories must possess FDA Food Facility Registration, designate a U.S. Agent, and hold cGMP certification; furthermore, they must complete product listing and labeling compliance reviews. Failure to meet these requirements will prevent customs clearance and product listing on Amazon.
2. Quality Control and Traceability Must Be Verifiable
- Raw materials require accompanying Certificates of Analysis (COAs). The production process must strictly control for microbial contamination, heavy metals, and prohibited ingredients. Every batch of finished product must be fully traceable to prevent product recalls or account suspensions resulting from quality issues.
3. Export Procedures and Liabilities Must Be Clearly Defined
- The factory must hold the necessary qualifications for exporting food products and cooperate in providing all required customs clearance documentation. Additionally, a contract manufacturing and confidentiality agreement must be executed to clearly delineate responsibilities regarding regulatory compliance, product quality, and intellectual property infringement.
If your supplier can’t provide all three upfront — before you pay a single dollar — that’s not a “yellow flag.” It’s a dealbreaker.
Why Honson Global Is Different
At Honson Global, we’ve spent 20 years building a vetted network of FDA-registered, GMP-certified manufacturers. Here’s what that means for you:
- Every manufacturer in our network has been personally audited by our team — not just verified on paper
- Every batch comes with a full COA report, tested by third-party labs
- Every product ships with complete compliance documentation — FDA registration, GMP cert, and ingredient traceability
- We’ve processed 15 million+ orders across 2,000+ products without a single compliance-related recall
You don’t have to become an FDA regulation expert. That’s our job.
Your job is building your brand. Our job is making sure nobody can shut it down.
Bottom Line
The supplement market rewards brands that get compliance right from day one. Not because it’s exciting — because it’s the difference between a brand that lasts and one that gets a warning letter in its first year.
If you’re about to place your first order — or if you’re already working with a supplier and you’re not 100% sure they’re actually compliant — let us check for you.
We’ll review your supplier’s documentation at no cost and tell you exactly where you stand. No sales pitch. Just the facts.
